

- 中文
- English
- Español
Diamonds for Life: Developments in Sensors for Biomolecules
2023-12-06 09:40:57
Click:
“
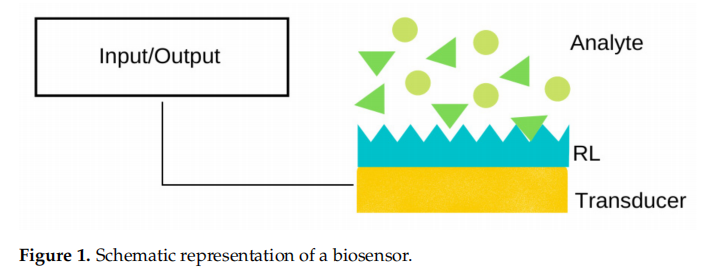
A biosensor is, generally speaking, a device composed of a receptor layer (RL) and a
transducer, as schematically depicted in Figure 1. Upon contact with a solution of the target
analyte (e.g., DNA, proteins, lipids, saccharides), it binds to the RL, and the transducer
typically translates the event into an output signal, such as an optical, thermal, piezoelectric,
electrochemical, or electrochemiluminescent signal, to name a few.
”
Diamond-based electrodes and biosensors are interesting in analytics because of their particular set of properties, namely: large potential window, chemical inertness, low baseline current, stability, and transparency. Diamond-based electrodes and biosensors were shown to detect biological molecules such as neurotransmitters and proteins, respectively. In this review, we summarise the different types of diamond electrodes and biosensors based on their type of detection (electrochemical or optical), functionalisation, and target analyte. The last section presents a discussion on the different analytical responses obtained with electrodes or biosensors, according to the type of analyte. Electrodes work quite well for detecting small molecules with redox properties, whereas biosensors are more suited for detecting molecules with a high molecular weight, such as DNA and proteins.

1、Introduction
A biosensor is, generally speaking, a device composed of a receptor layer (RL) and a transducer, as schematically depicted in Figure 1. Upon contact with a solution of the target analyte (e.g., DNA, proteins, lipids, saccharides), it binds to the RL, and the transducer typically translates the event into an output signal, such as an optical, thermal, piezoelectric, electrochemical, or electrochemiluminescent signal, to name a few.
Diamond gathers a unique set of chemical and electronic properties that make it aquite a promising material for the composition of the transducer layer of a biosensor Chemical inertness makes diamond thin films safe to use in biological applications while the inherent hardness and mechanical resistance of diamond allow diamond-basedsensors to be very durable and to operate under aggressive conditions such as high or low pH media.
Natural diamond is an insulator with a very wide band gap ( 5.5 eV) and anexceedingly high electrical resistivity (1016 0cm), hindering its application in electronics. In turn, synthetic diamond deposited by chemical vapour deposition (CVD) in hydrogen-saturated conditions presents a much lower surface resistivity, with a value around 10% 0cm. The conductivity of diamond can be further tailored by including lattice-selected impurities, such as boron, nitrogen, and phosphorous, during the CVD process.
The target-specific receptor layer depicted in Figure 1 can be achieved by the functionalisation of hydrogen-terminated CVD diamond films with amino or carboxylic pending moieties capable of linking a biomolecule. Photochemical methods (e.g., alkenes) or electrochemical modifications (via reduction of diazonium salts) are the most common methods to attach these pending groups to the diamond surface. This is followed by a secondreactional step, where a complementary biomolecule (to the target analyte) is attached to these linkers, thus conveying high sensitivity and specificity to the diamond layer.This review describes the main methods of fabricating diamond-based biosensors and is organised as follows: Section 2 describes the characteristics of diamond films deposited by CVD and the functionalisation of their surface using plasma treatments; Section 3presents an overview of the photochemical, electrochemical, and green chemistry methods used in the surface modification of diamond films; Sections 4 and 5 describe the practical applications of diamond-based electrochemical sensors and of biosensors with a diamond receptor layer, respectively. We note that the sensing devices are listed according to the type of analyte, with electrodes finding applications in the analysis of small, redox-activebiomolecules such as histamine, dopamine and other neurotransmitters, and biosensors with a larger surface area being used on the recognition of biomolecules such as DNA and proteins. At the end of the review, a comparison weighing the benefits and drawbacks of these various types of detectors is presented.
2、Chemical Vapour Deposited Diamond Thin Films
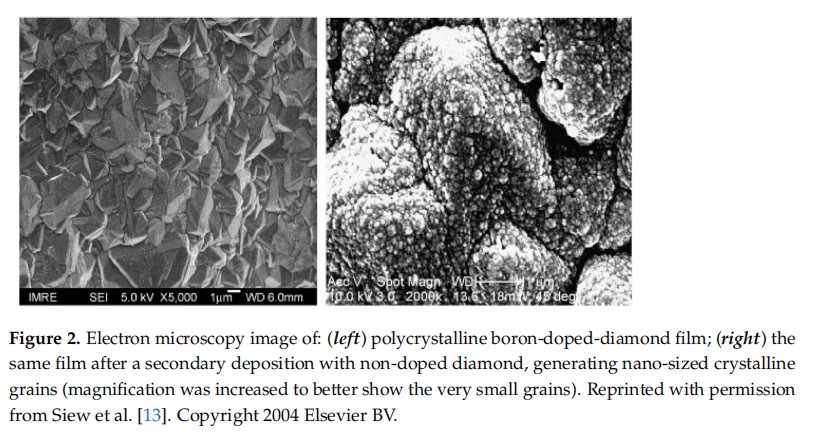
Diamond thin films can be deposited by CVD on a variety of non-diamond substratesin a carbon-containing atmosphere saturated with hydrogen. The latter plays a pivotal role: on the one hand, hydrogen atoms generate radicals on both the hydrocarbon source and the diamond substrate; on the other, they etch sp--hybridised carbon (graphite) away at a much higher rate than sp2-hybridised carbon (diamond), which results in the growth of high-quality diamond films. Hydrogen atoms further stabilise the dangling bonds at the diamond surface, being at the genesis of some of the most unique properties of diamond, such as high chemical inertness, large potential window, hydrophobicity, andnegative electron affinity (NE A).Natural diamond is an insulator. When films are deposited by CVD in a hydrogen-richenvironment, they exhibit a property deemed surface conductivity. This happens because carbon is slightly more electronegative than hydrogen, resulting in the formation of a dense surface dipole layer with slightly negative charged carbon atoms (C-) and slightly positive charged hydrogen atoms (H+), which causes an electrostatic potential step AV perpendicular to the surface over a distance of the order of the C-H bond length, ie, 1.1 A Energy variation over this dipole is in the range of 1.6 eV. The conductivity of diamond can be further tailored by including charge-carrierelements, such as boron, nitrogen, and phosphorous, into its lattice. Doping diamond with boron makes it become a p-type semiconductor, with an activation energy (Ea) of 0.37 eV while n-type behaviour can be obtained by doping diamond with nitrogen, Ea = 1.7 eV, or phosphorous, E,= 0.56 eV.The surface of a diamond thin film is typically polycrystalline, with grain sizes within the micrometric range. The versatility of the CVD technique allows for the preparation ofmulti-layered materials, where doped and undoped layers can be deposited one on topof each other simply by changing the composition of the gas environment, allowing the fabrication of films with tailored properties. An as example, the electrical properties of the material stack can be improved by growing a thin layer of non-doped nanocrystalline diamond on top of a boron-doped polycrystalline film (Figure 2).
When the undoped nanolayer is deposited over a base layer of conducting polycrystalline diamond, the combined material can be employed as an electrode with improved selectivity for small biomolecules. A literature example reports the deposition of nanocrys.talline diamond over boron-doped diamond using two hours of deposition at 20 Torr50 sccm CH4, 200 sccm H, 2 sccm C,H, a substrate bias of -200 V, and a substrate temperature of 800 °C) .More recently, Song et al. showed that it is possible to produce boron-doped diamond(BDD) starting from non-doped diamond and using a secondary deposition to perform the boron doping. As expected, BDD samples presented electrochemical properties that the diamond film did not have. As the thickness of the doped layer increased (by increasing the time of doping), there was an increase in the potential window, but there was no change in electrochemical activation, because this is a surface phenomenon.
Plasma Treatment of Diamond Thin Films
The surface of a diamond thin film typically bears hydrogen atoms (H-diamond). It is possible, nevertheless, to produce diamond films with other terminal groups, influencing in this way, properties such as electron affinity, surface conductivity and wettability andultimately changing its ability to adsorb and attach molecules, cells, and bacteria Oxygen and nitrogen are two of the most common terminal groups of diamond surfaces This termination can be achieved by exposing the diamond to selected gas atmospheres under conditions that allow them to react with the diamond surface.The replacement of H atoms with O ones can be achieved by exposure to a plasma of oxygen, or to atomic oxygen generated from a radio frequency (RF) plasma or by thermal oxidation in oxygen atmosphere. Unlike the original H-diamond the surface of diamond, being rich in oxygen, is hydrophilic, and has a positive electron affinity (PEA). Its electrochemical properties (that is, electron transfer kinetics and potential window) are different from those of the surface of H-diamond.While the existence of oxygen terminal atoms guarantees a hydrophilic surface, the presence of nitrogen ones brings the additional advantage of allowing the covalent binding of biomolecules such as DNA and peptides. In a similar fashion to the oxidation process, the inclusion of nitrogen atoms onto the diamond surface can be achieved with an RF plasma treatment in an atmosphere of He/NH3 or in N2. The latter process can generate multiple bonding configurations, such as C=N, C-N, and C-NH2.
3、Surface Modifcation of Diamond Thin Films
In addition to the plasma reatment, specific molecules can be altached to the surface of the diamond films using a variety of techniques, such as photochemical and electrochemicafunctionalisation. "Greener" chemical methods have been further reported in the literature
3.1. Photochemical Renction
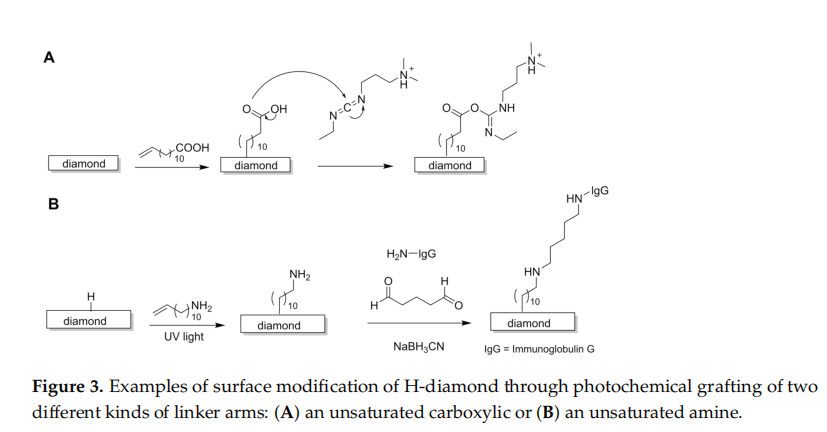
One of the first methods for the inclusion of different functional groups onto thesurface of H-diamond resorted to processes initiated by photochemistry. In these, UVlight is employed to attach simple NH groups or bridging molecules, such as acid oramine-terminated alkenes, to the surface of the diamond film (Figure 2). The bridging molecules create a spacer arm that can subsequently form a covalent bond with a target specific molecule, such as a DNA oligonucleotide chain or a protein. Moreover, surface modification by photochemical methods has advantages over wet-chemical modification because it allows a facile patterning of the substrate with functional groups. As a setback the presence of two different functional groups in the amino alkenes, e.g., a double bond atone end and an amine at the other end, requires the use of protecting groups to ensure that the photochemical reaction occurs with the double bond site and to avoid reaction of the amine head groups, which may cause etching of the substrate.
The simplest form of functionalisation involves the generation of Nho-terminatecsurfaces in diamond. These can be obtained by the exposure of H-diamond to chlorine gas under irradiation with a 450 W Hg arc lamp (254 nm), with subsequent exposure of the chlorine-terminated to ammonia gas, also under irradiation. Both reactions are conductedat low pressure (c.a, 10-7 atm)
Methods using spacer arms are preferred for a better spatial accommodation of largemolecules such as DNA. The first report on the functionalisation of nanocrystalline diamondfilms with a DNA oligonucleotide sequence dates back to 2002 and employed 10-aminodecl-ene (with the amine group being protected by trifluoroacetamide) to form a covalent bond with diamond carbons by irradiation with UV light (0.35 mW cm-2, 254 nm). After deprotection, the primary amine was allowed to react with a heterobifunctional crosslinkersulfosuccinimidvl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (SSMCC), to form agroup that subsequently reacted with a thiol-modified DNA oligonucleotide chain.
Alternatively DNA oligonucleotide chains can be linked to H-diamond by an alkenebridge terminated with a carboxylic acid group at the other end. A commonly em.ploved linker is 10-undecenoic acid, which reacts by activation with UV irradiation (20 h2.5 mW cm-2, 254 nm) to form the bridging layer; subsequently, the free carboxylate groupsare treated with 1-ethyl-3-[3-dimethylaminopropyl]-carbodiimide (EDC), generating anactive intermediate species that readily reacts with the amine groups of the desired DNAoligonucleotides (Figure 3A). Activation of the carboxylate groups with EDC permits a much faster, high-vield reaction because the active intermediate is more reactivethan the native COOH. Further development of this method compared the effect of reacting10-undecenoic acid in the unprotected and protected forms (trifluoroethyl undecenoate)having shown that the use of protecting groups leads to a lower density of carboxylategroups on the surface of diamond. This is likely due to the bulkier nature of the pro.tected molecules that create a steric hindrance to the functionalisation reactions, reducing the density of the attached molecules.
The inclusion of an antibody to the surface of H-diamond followed a process similatto that already described for DNA, in which trifluoroacetamide-protected 10-aminodec-ene was employed in the first reaction step (Figure 3B). In a second step, the substratewas trealed with a solution of glutaraldehyde, a protein cross-linking agent able to reactwith amines to form Schiff bases. This method required, however, an extra treat-ment step (addition of 0.1 M solution of glycine in sodium cyanoborohydride couplingbuffer) to remove unreacled aldehyde groups that could affect the slability of the surface of diamond.
3.2. Electrochemical Reactions
There procedures are based on the reaction of charged molecules with the surface ofH-diamond through the use of an electric current.
3.2.1. Reactions Involving Diazonium Salts
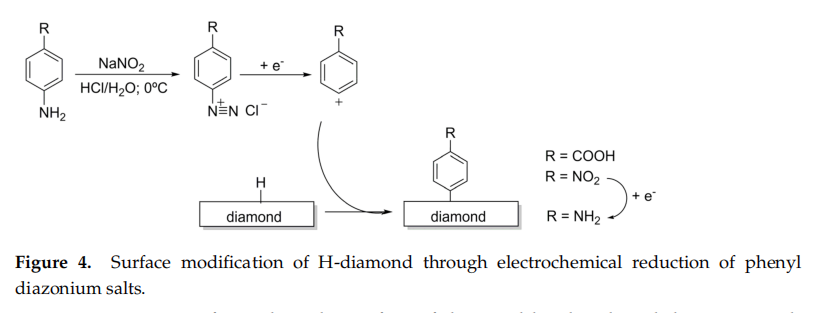
The use of phenyl diazonium salts was first reported by Kuo et al. in 1999 andquickly became an extensively employed method for the functionalisation of diamond surfaces with linker arms that serve as anchors for the covalent bonding of biomolecules suchas antibodies or DNA oligonucleotide chains. The process starts with the synthes is of a diazonium salt, e.g., 4-nitrobenzenediazonium tetrafluoroborate (Figure 4) , whichis then used to treat H-terminated diamond thin films (40 mM) in the presence of a solution of 1% SDS (sodium dodecyl sulfate) to produce a surface terminated with 4-nitro-ary!groups. Subsequent application of the current reduces the nitro groups to primary amines.
Primary amines formed on the surface of diamond by the phenyl diazonium saltmethod can be used as anchoring points for a variety of molecules. An example of this is the reaction with thiol-modified DNA oligonucleotides via SSMCC cross-linking (details of the process for cross linking DNA with NH-terminated diamond are describeo in Section 3.1).A drawback of this method is when the phenyl radical species react not only with thesurface of the diamond film, but with the phenyl ring grafted on the surface, ultimately forming undesirable multilayers.
3.2.2. In Situ Electrochemical Polymerisation
Electrochemical reactions can be fine-tuned to achieve in situ polymer deposition. An example of this reported the use of pyrrole, a molecule with a positive charge, to form apolypyrrole film: the diamond surface was immersed in a solution of 50 mM pyrrole in 0.2 M KCl and cycled twice from 0 to 1.1 V (sweep rate of 500 mV s-'). Subsequentoxidation (by adding 0.5 M NaOH and applying ten electrical cycles), promoted theformation of COO- groups, resulting in a polymer coating with multiple negative charges.This method was reported to improve the detection of small biologic cationic molecules such as dopamine.
3.3. Reactions Promoted by lonic Liquids
Most of the methods reported so far rely on the use of an external energy source(RF/ thermal/electrical energy of UV light) to provide the required activation energy to establish the bonds between the atoms on the diamond surface and the different chemicalentities. A new perspective for the use of diazonium salts in the chemical modification of the surface of diamond films bearing hydrogen atoms has been proposed and uses ionicliquids. The process can be conducted without the use of any source of activation that is, without thermal, electrical, or photochemical activation. In their report, Szuneritsetal. used only 4-nitroazobenzene dissolved in ionic liquids, taking advantage of their low vapour pressure: droplets of an ionic liquid solution of the diazonium salt are deposited locally on carbon materials by using simple spotting techniques, opening the way for new opportunities in the chemical functionalisation of diamond surfaces.
4、Diamond-Based Electrodes
Boron-doped diamond (BDD) electrodes are increasingly employed in bioanalyticalapplications because of their unique electrochemical properties, such as a low and stable baseline current and wide potential window of water decomposition. Chemicalstability is a key factor: while most of the commonly emploved electrodes cannot be use din corrosive, acidic, or basic media, the chemicalinertness of diamond electrodes allows for their use in every type of media. Another benefit is the 'anti-fouling' behaviour, i.e.their poor adsorption of most types of organic molecules, which ensures a very good stability against the degradation of the electrochemical signal over time due to surface passive layer build-up.
Diamond-based electrodes are reported to detect a variety of biogenic amines, including neurotransmitters such as dopamine, adrenalin, noradrenalin, serotonin , and histamine. Detection of this class of molecules is typically based on their oxidation reactions, in which the alcohol moieties of the catechol are transformedinto ketones. Electrodes described in this section are typically not functionalised fortarget-specific analysis, and their sensitivity is rather attained by finding the oxidation (or reduction) peak voltage of the desired analyte and then operating in amperometric mode.that is, fixing the voltage at the previously determined optimal value. Increased sensitivity can be achieved with simple coating procedures, such as using an oxidised polypyrrolepolymer to repel anionic contaminants, or by reshaping the electrode surface with nano-grains to narrow the redox peak of a target analyte. Specificity can be increased by coupling the electrode with a separation method such as capillary electrophoresis or flow injection analysis.
4.1. Histamine Detection
The electrochemical response of a polycrystalline, BDD thin-film electrode to histaminein aqueous media at a pH of 7.2 was investigated with cyclic voltammetry and hydro.dynamic voltammetry. The cyclic voltammograms of histamine were well definedpeaking at 1.40 V, having a detection limit of 1 µM and a linear response range for his tamineconcentrations between 0.5 and 100 ul.
4.2. Serotonin Detection
The same BDD thin films described in the previous subsection were applied to the detection of serotonin. Cyclic voltammograms showed well-defined sweeps, with peaks indicating no adsorption of the oxidation products of serotonin on the surface of the electrode. Consistently no fouling or deactivation of the electrode was observed within the entire length of the experiment (several hours). The linear dynamic range was within the 0.01 to 100 µM interval with a remarkably low limit of detection around 10 nM.
4.3. Dopamine Detection
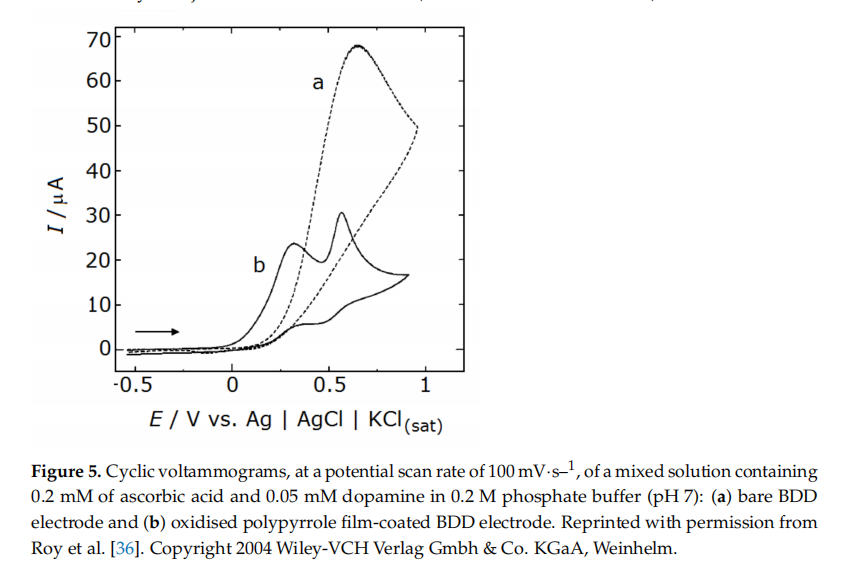
Voltammetric detection of dopamine was achieved with a micro-electrode preparedfrom a BDD film and modified by a secondary deposition process (see details in Section 2to exhibit an external layer of nano-sized undoped crystalline diamond. This strategy helped to narrow the oxidation peak of dopamine, making it discernible from the signals of two common interferents, ascorbic acid and uric acid. Note that, when the diamond electrode was not treated (with the second deposition) to feature the nanocrystals, the peaks of these two interferents overlapped with that of dopamine.An alternative strategy to detect dopamine in solutions with other contaminantsresorted to a BDD electrode deposited on a microfiber and coated with a polymeric layer of oxidised polypyrrole (see manufacturing details in Section 3.2.2). The presence of the oxidised polypyrrole coating helped to reduce the interference originated from ascorbicacid and 3,4-dihydroxyphenylacetic acid (DOPAC), a common metabolite of dopamine. This behaviour is associated with the abundance of negatively charged carboxylate groups from the oxidised polypyrrole coating) on the electrode surface that attracts dopaminecations while repelling the negatively charged ascorbate anions. Even when an excess of ascorbic acid (0.2 mM) was present in regard to the target analyte, dopamine (0.05 mM)voltammograms showed two separate peaks, one for dopamine at 0.5 V and another forascorbate at around 0.3 V (Figure 5) The limit of detection for dopamine was 0.1 nMatS/N = 3 (S/N is the signal-to-noise ratio), and the linear dynamic range laid between0.5 nM and 100 uM. The amperometric response for 0.5 nM dopamine showed high stability relative standard deviation of 5.4% for n = 5) and good reproducibility (within ± 6.2% forn = 10, with a solution of 1 nM of dopamine). Furthermore, the response remained the same during measurements conducted over a period of seven days, with no variation inthe efficiency of rejection of the interferents (ascorbic acid and DOPAC).
Sensitivity to dopamine in matrices containing complex backgrounds can be further increased using coupled systems. In one of such report, amperometric detection of dopamine using a BDD microelectrode was coupled with a pretreatment using flow injection analysis. The system detected dopamine with good reliability and reproducibilityat concentrations as low as 6.0 µM (the lowest tested concentration) even when the analytewas dissolved in complex matrices such as river water and human serum. In another reportelectrochemical detection of dopamine using a BDD microelectrode was coupled withcapillary electrophoresis. The system allowed the separation and analysis of dopaminein a mixture containing dopamine, catechol, and ascorbic acid (all at concentrations of10 mM in phosphate buffer at pH = 6). The system performance was evaluated at differentapplied potentials, showing that the background current (~100 pA) was very stable with time and good reproducibility of the times of elution and detection (peak current or area)for each of the components of the mixture. The response had a precision better than 4.1%, a linear response between 0.1 and 1 mM, and a limit of detection of 1.7 fM.
4.4. Multi-Component Analysis: Dopamine, Adrenaline and Noradrenaline
A similar setting to that previously described, that is, comprised of a boron-dopedmicroelectrode coupled with capillary electrophoresis, was reported to simultaneouslydetect three main neurotransmitters: dopamine, adrenaline, and noradrenaline. Thesystem exhibited high separation efficiency for these three catecholamines and a good detection performance, with high reproducibility for the current response over ten repetitiveiniections of mixtures of the catecholamines, at 50 µM each, and detection limits as low as20 nM.
4.5. Tounrds In Vivo Applications: Noradrenaline and Adenosine Detection in Excised Tissue
4.5.1. Noradrenaline
In an attempt to bring sensors closer to real-life applications in living organismsa diamond-coated carbon fibre microsensor was developed and tested regarding noradrenalin detection in an in vitro setting with excised rat mesenteric tissue. Followingelectrical stimulation of the mesenterium (60 pulses at 20 Hz for a total time of 3 s, repeating the stimulation after 10 min), noradrenaline was released from sympathetic nerves innervating the mesenteric artery and was measured as an electric response, presenting a peak of c.a. 10 pA. The response exhibited good reproducibility and recyclability over time.
4.5.2. Adenosine
Adenosine, besides being a base component of DNA, is present in the brain at lowconcentrations (around 50 to 200 nM), participating in neurotransmission modulationparticularly in the control of respiratory function. In a pilot study, Xie et al. focused on thePreBotzinger complex in rodent brains, a site thatis rich in adenosine-releasing neurons. Excised sections of this brain region were maintained in artificial cerebrospinal fluid (with8 mM K+). Measurements resorted to a BDD thin film grown on the tip of a tungstenmicroelectrode (with an approximate diameter of 30 µm) that was shown to detect aconcentration of 10 nM of adenosine and had a signal-to-noise ratio of 20 with a limit ofdetection of 2 nM(S/N = 3). When placed over the excised brain tissue slice stimulated with a synthetic drug to release adenosine, the microelectrode was able to detect the adenosinerelease in concentrations oscillating between 2 and 5 µM.
4.6. Detection of NADH, a Dinucleotide of Adenine and Nicotinamide
Nicotinamide adenine dinucleotide (NADH) is composed of two nucleotides linkedthrough their phosphate groups. This essential biomolecule for cell metabolism is involvedin a variety of cellular redox reactions, including the citric acid cycle and the oxidative phosphorylation chain, two essential processes for energy production in living cells. Detectionof NADH was achieved with a BDD thin film electrode, with well-defined voltammogramseven at low NADH concentrations (0.2 mM), with a good amperometric performance anda detection limit of 10 nM (S/N ratio of 7). The electrode response was found to be stablefor a storage period of up to 3 months.
4.7. A Glutathione Electrode Operating In Vivo
Glutathione, a redox-active tripeptide with the sequence L-y-glutamyl-L-cysteinylglycine, is the main biomolecule responsible for the removal of oxidising species and frecradicals that can cause damage to living tissue. A BDD was reported to detect glutathioneboth in chimico and mboxin vivo in a mouse model. Amperometric measurementsconducted in solutions of glutathione in diluted phosphate buffer saline (PBS) showeda limit of detection of 0.3 mM for glutathione at S/N = 3. In vivo measurements wereconducted subcutaneously in immunodeficient mice with xenograft tumours derived fromhuman cancer cells. Glutathione is typically overexpressed in tumour cells due to their augmented metabolic rate. The electrode was shown to detect the difference in concentrationsof glutathione between cancerous and healthy tissues with high reproducibility.
5、Diamond-Based Electric Biosensors
The possibility of functionalising the surface of the diamond films with a specific detector layer opens the door to the detection of larger and specific molecules such as DNA or even viruses. The operating principle of these biosensors is based on the occurrence of intermolecular interactions between the target molecules and the receptor layer, which bring them improved selectivity. The use of more than one type of receptor molecule on the surface allows for the production of sensors capable of detecting multiple analytes. In addition, when molecules of the analyte are recognized by their counterparts at the receptor layer, the supramolecular interaction results in a change in the electrical properties of the surface of the biosensor that allows for other interrogation techniques in addition to cyclic voltammetry, such as impedance measurements. By specifically probing the changes in the impedance of the surface, these measurements may provide a more accurate tool to detect the presence of the molecules of the analyte.
5.1. Biosensors for DNA
Yang et al. observed the recognition of DNA oligonucleotides at the surface of diamondthin films by frequency-dependent impedance measurements. Measurements as afunction of potential show that the impedance at 106 Hz was dominated by the spacecharge region of the diamond film and that the hybridisation of a complementary DNAoligonucleotide was able to induce a field effect in the diamond space-charge layer. Thiseffect altered the impedance of the diamond film, thus allowing for a real-time hybridisationdependent response.Zhong et al. tested the electrical detection of DNA using two types of diamondthin films, one composed of non-doped diamond and another of BDD with dopant concentrations above 1019 cm-1. Both films were functionalised with undecenoic acid with subsequent attachment of a DNA probe chain. Interestingly, monitoring DNA recognition by impedance measurements demonstrated that there was an insignificant change in impedance following hybridisation of DNA at the BDD-based biosensor, while theDNA sensor built from non-doped diamond exhibited a clear reduction in the impedanceresponse. This was explained as the result of 'reduced charge-transfer resistance of the molecular layers following the 7t-nt stacking of the duplex DNA, as well as a reduction in the subsurface depletion due to an accumulation of holes.A DNA impedimetric biosensor built from undecenoic acid/DNA-functionalisednon-doped nanocrystalline diamond films was reported by Vermeeren et al. This device operated in a similar way to that constructed by Yang et al, that is, it showed adecrease in impedance when the complementary target DNA was added. Further test sincluded challenging the biosensor with a solution containing a DNA sequence that was not complementary to that of the receptor layer (with a single nucleotide mismatch, or 1mismatch) to investigate if it could be differentiated from the target analyte. Results showed that differentiation between 1-mismatch and complementary target DNA was possibleat frequencies between 100 and 1500 Hz. The biosensor still showed, however, some limitations for a wide range of applications, because its performance suffered negatively from the low ionic content of the sample media. Hybridisation in 1x PBS buffer wasunsuccessful, while the separation between complementary and 1-mismatch hybridisationcurves was very clear and reproducible when using the concentrated buffer, 10x PCR buffer (at 30 °C).
5.2. Biosensors for lmmunoglobulins
Detection of antigen-antibody recognition using a biosensor built from a diamondthin film was first reported in 2007, resorting to electric impedance measurements of theimmunoglobulin G /anti-immunoglobulin G antigen-antibody system (29].A biosensor for immunoglobulin E (lgE) was subsequently reported by Tran et al. (55nstead of using the native anti-lgE antibody, an alternative approach was employedwhich resorted to a smaller, synthetic biomolecule called an aptamer for the receptor layerbridged via undecenoic acid). Aptamers are oligonucleotide or peptide chains designedto recognise DNA or proteins. The aptamerwas a synthetic chain of oligonucleotidesenriched with a terminal sequence of twenty thymidine bases for maximum flexibilitin binding to the lgE protein. The biosensor offered a low detection limit of 0.03 ug/ mlat S/N = 3) and a linear dynamic range between 0.03 and 42.8 ug/ mL. Reusability wastested for six cycles, with relative standard deviations (after the sixth cycle) rounding to%. The practical utility of the biosensor was tested in human serum samples, with result.showing a linear dose-response for concentrations of lgE up to 5.8 ug /mL and an excelenicorrelation coefficient of 0.99 between quantification data provided by the biosensor andthe reference method , enzyme-linked immunosorbent assay (ELISA)
5.3. Biosensors for Influenza
5.3.1. Detecting M1 Protein as a Universal Influenza Biomarker
Influenza A virus matrix protein M1 is a widely abundant membrane-associatecprotein that lies under the envelope of the virus particles, helping to maintain structuraintegrity and participating in several essential processes of the viral life cycle. When released into the serum, M1 proteins can agglomerate and help the various components of the virus assemble to form a new viral particle. M1 can be monitored as a biomarker of influenza infection with an important advantage over other antigen proteins because it is transversal to all serotypes of influenza virus.A biosensor for the M1 protein of influenza was built from a nanocrystalline BD Diamond thin film functonalised with polyclonal antibodies yia diazonium salts electrodeposition. The biosensor was able to detect the M1 protein in the H1N1 in.luenza strain by impedance measurements in PBS supplemented with 0.5% Triton Xa non-ionic surfactant. Under these conditions, the sensor response stabilised within5 min, presenting a linear range of 10 to 100 fg/mL and a limit of detection as low as0.7 fg/mL, which corresponds to approximately 40 viral particles. The biosensorwas also able to detect the H3N2 strain under the same conditions with almost no changein the impedimetric response.
5.3.2. Detecting Whole Influenza Viral Particles via Hemagglutinin (HA) Recognition
A biosensor for whole viral particles of influenza was built from a BDD thin filmfunctionalised by the electrochemical deposition of a diazonium salt with the subsequentattachment of a sialic acid mimic dimeric peptide. This peptide was designed for therecognition of hemagglutinin (HA), one of the surface antigens of the influenza virus. Thefirst sequence of tests demonstrated that the biosensor does recognize pure HA in a PBS solution when operating in the mode of electric impedance spectroscopy. The biosensor was tested with whole viral particles, having proven successful in responding to botlH1N1 and H3N2 strains in the concentration range of 400-8000 pfu/mL (where pfu is a plaque-forming unit).
6、Discussion and Future Outlook
The electrical detection of biomolecules using diamond-based devices can be done either with simple electrodes or with target-specific biosensors, as reviewed in the present work.Electrodes built from BDD (Section 4) can serve as practical, resilient, and reusableinstruments in bioanalytical applications because of their unique electrochemical propertieschemical stability, and "anti-fouling' behaviour. Unlike the biosensors described in Section 5these electrodes are not functionalised for target-specific analysis, their optimisation beingattained by finding the oxidation (or reduction) peak voltage of the desired analyte ancoperating under that voltage. This raises the obvious question of selectivity, particularly for the electrodes described in Sections 4.1-4.4, which were only evaluated in chimico, that is, irbuffered solutions containing either just the target analyte or mixtures of the analyte withone or two common interferents. Their ability to operate in complex biological matricessuch as biological fluids or even in a living tissue, remains to be demonstrated, being an important target of investigation for those aming to pursue further studies in this field. As econd important issue is their sensitivity (LOD and/or linearity range), because biologicamines exist in very low quantities in the human body. Plasma levels of dopamine range between 0.05 and 0.3 nM, while plasma noradrenaline is within 0.9 to 8 nM. The sevalues are below, or within close proximity of, the linear response range of the electrodes thus meaning that their applicability to real-case scenarios with plasma samples will requirea pretreatment step to increase the concentration of the analyte, or the improvement ottheir performance. The approach employed by Roy et al. is a possible strategy for curbing these issues: by coating the electrode surface with an anionic polymer, the LOD was successfully lowered to the fentomolar range; however, the linear response range was still quite high, within the micromolar range, warranting further improvement. A recentstrategy to overcome reduced sensitivity was recently presented by de Brosse et al. 60Using an oil-membrane composite protection chamber, interferences from the biofluid othydrophilic solutes and solvents were mitigated. This oil-membrane protection allowedowering the LOD of two standard inorganic compounds, hexacyanoferrate (ll/Ill) anchexaammineruthenium (ll/1ll), by over 100-fold compared to traditional electrodes (that is, those that were not covered with the membrane/oil chamber). While this concept was only demonstrated for the two inorganic standards, it is an innovative and promising approach for detecting biomolecules in biofluids with fewer performance-diminishinginterferent effects; moreover, it does not require the change of the biosensor system its elfrelying, instead, on the use of a careful selection of the semi-permeable membrane and of composition for each specific application.Electrodes described in Sections 4.5-4.7 constitute interesting proofs-of-concept on the application of diamond-based devices for sensing biological transmitters. The adenosine electrode, tested by Xie et al. on excised rat brain tissue and shown to operatewithin the nanomolar range (5l], may also find use in monitoring plasma adenosine level stypically around 0.6-1.1 mM. The NADH sensor (Section 4.6) also operates in the nanomolar range. Further emphasis should be given to the electrode reposted by Fierroet al. (Section 4.7), already demonstrated to operate in vivo with promising results incancer detection.
In the case of diamond-based biosensors, specificity of response is implemented by design, because the response relies on a specific interaction between the analyte and acomplementary biomolecule on the organic receptor layer. The supramolecular recognition occurring between these two biomolecules, the covalently bonded one at the RL and the target analyte, creates a high degree of selectivity, which cannot be matched by electrodes.An additional advantage of biosensors is that they can operate with very small amounts of sample: a droplet of the solution can be simply placed on the receptor layer of the diamond-based biosensor. Furthermore, carbon-made biosensors with electrochemical detection have already been used to detect highly proliferative cells, such as human breast adenocarcinomaMDA-MB-231), breast cancer isolated cell line (MCF-7), and hepatocellular carcinomaisolated cell line (HepG2), as portrayed and summarized by Suhito et al., showing a great potential of carbon-based materials not only to detect biomolecules, but also the ability to provide a label-free, non-invasive, and non-destructive method for detectingearly-stage cancer. This could be an interesting approach for boron-doped diamond in the future.
In conclusion, when weighing diamond-based electrodes against biosensors in the detection of biomolecules, there are, in one hand, the practical advantages of the easy-toproduce and easy-to use diamond-based electrodes and, on the other hand, the outstanding ability of biosensors to detect biological molecules of high molecular weight with a lower amount of analyte and very high selectivity. The manufacturing of biosensors require show ever, a sequence of chemical functionalisation steps, which may pose as a disadvantage because of the increased time, cost, and complexity of fabrication. For these reasons, the choice between a diamond-based electrode and a biosensor depends on several issues!the size, charge, and solution mobility of the target molecule; the amount of analyte; the intended type of detection; the intended selectivity and the time and money for fabrication.

-

Z-Axis System: The Key to Stable Production of MPCVD Diamonds
Microwave Plasma Chemical Vapor Deposition is the mainstream method for producing high-quality diamonds in the industry. When the equipment is running, it first pumps the reaction chamber into a vacuum state to strictly ensure the chamber is fully sealed and airtight. Then, the microwave device releases energy, and special mixed reaction gases are fed into the chamber. Under the action of energy, a stable plasma area is formed. These highly active plasmas provide stable heat and reaction conditi
-

How to Choose the Right MPCVD Equipment?
MPCVD equipment is a critical asset across a wide range of industries, including industrial manufacturing, semiconductor thermal management, precision cutting tools, and lab-grown diamonds. Choosing the right equipment not only boosts production efficiency and ensures consistent product quality but also effectively controls long-term costs, making your entire production process smoother and more competitive. Many people struggle with the question: How do I make the right choice? Today, drawing o
-

CVD Diamond Clarity: How to Avoid Inclusions That Affect Jewelry Value
After selecting a loose diamond, the setting not only determines the diamond’s security but also directly affects its perceived size. With diamonds of the same weight, choosing the right setting design can effortlessly create the surprise of “small carats making a big impact,” saving on your budget while enhancing the diamond’s quality. Among these, pavé and prong settings are the two most classic and effective ways to make a diamond appear larger. Paired with specialized designs that maximize t
-

How Diamond Became the Ultimate Thermal Solution in the Era of Computing Power
The iconic narrative behind the legendary "A Diamond is Forever" slogan has long been upended by China's industrial prowess. Once synonymous with luxury and rarity, this gem has shed its glamorous aura to become an indispensable thermal management material for high-power devices. In an era of explosive growth in AI computing power, it has taken up the mantle of "cooling to save computing power".










